NM23 Media© ®™
NM23 minimal media functions similarly to bFGF, with our novel NM23 growth factor being added directly to stem cell media. This allows for growth and maintenance of large populations of undifferentiated human stem cells with the added benefit of being used in a completely defined feeder-free system making it ideal for therapeutic purposes.
Add 8 nM rNM23-RS to Minimal Media:
- DMEM/F12 media
- Knockout Serum Replacement
- Non-Essential Amino Acids
- Beta-Mercapto-Ethanol
Do not add bFGF or any other growth factor
Surface Coating
Human undifferentiated stem cells express the MUC1* growth factor receptor. Therefore, we can get specific attachment of undifferentiated human stem cells by coating anti-MUC1* antibody onto cell culture plate at 25 ug/mL. Incubate antibody with plate at 4oC O/N or at RT 3 hrs. For best results use Vita © ®™ plates.
The surface coating also provides user with a chemically defined and xeno-free surfaces that supports human stem cell growth combined with a feeder-free media using our novel NM23 growth factor.
Steps
Pluripotent stem cells can easily be transitioned to grow in NM23 media with minimal acclimatization:
- Stem cell colonies grown by the standard method (i.e with bFGF on MEF feeder cells) are initially washed with 8 nM rNM23-RS before being manually dissected in the continued presence of 8 nM rNM23-RS
- Cells are trypsinized for 3 min, 37oC, 5% CO2
- Following trypsinization, cells are incubated for 30 min in 8 nM rNM23-RS, 37oC, 5% CO2, during which time cells are counted in a hematocytometer.
- Stem cells are plated onto anti-MUC1* antibody coated 6-well cell culture plates in the presence of ROC inhibitor (EMD #688000) to final concentration of 10 μM.
- Cell plating density for initial transition to NM23 media is ~400,000 cells/well of a 6-well cell culture plate, then reduced to 200,000 by P3. Note: Optimal plating density can be assessed per cell line.
NM23 is Required & Sufficient for Pluripotent Growth
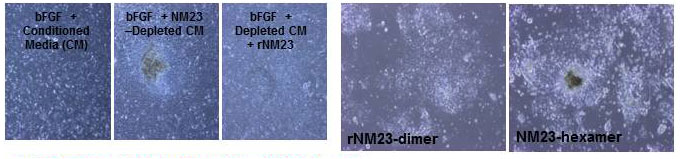
NM23 is the active ingredient in conditioned media:
MEF conditioned media does not support undifferentiated stem cell growth if it has been depleted of NM23. After reconstitution of conditioned media with recombinant NM23, the need for bFGF is eliminated. Why? Because wild type NM23 quickly goes to the hexamer form, which is inactive. Minerva’s NM23 is a constitutively active form that is always a dimer, which is the active form. Competitive inhibition of the NM23-MUC1* interaction with the free MUC1* peptide or the anti-MUC1* Fab induces differentiation and a spike in miR-145 which signals the cell’s exit from pluripotency.
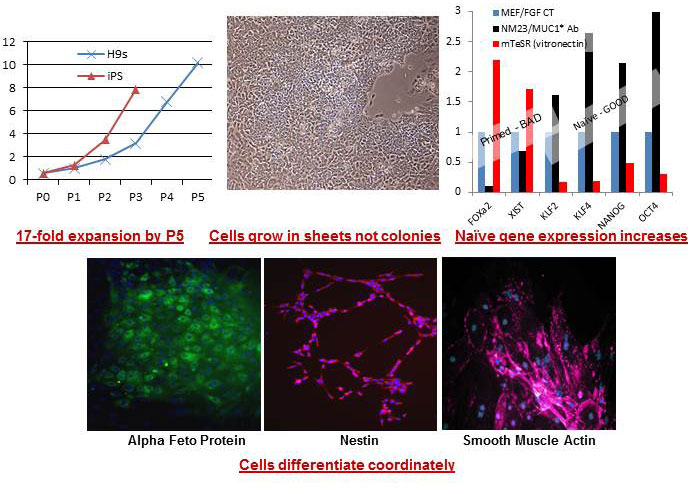
STEM CELL GROWTH
We believe we have discovered the one necessary and sufficient growth factor for human ES and iPS stem cells. Our stem cell growth factors are the only factors that do not stimulate the FGF pathway, which appears to convert stem cells from the "naÑ—ve" state to the "primed" state. We have also developed chemically defined and xeno-free surfaces that support human stem cell growth.
Over the past two years, we have developed an animal-free, cell-free system, comprised only of defined components, that enables massive growth of pluripotent stem cells, including iPS (induced pluripotent stem) cells, which are the patients own skin cells that are induced to revert to the stem cell state. We have also developed methods and a complete product line for automated, high throughput growth, identification and harvesting of stem cells.
This product line of stem cell growth factors represent a watershed advance for stem cell culturing. These new growth factors target a primal growth receptor, MUC1*. Through successful manipulation of this receptor alone we can keep stem cells in their pluripotent state, and passage them in animal-free culture conditions for many generations. Additionally, these new growth factors preclude the need for the complicated cocktail of less important growth factors and chemicals currently required to prolong pluripotency. By both simplifying and enhancing the growth conditions will allow scientists to grow more stem cells more reliably, for greater passage numbers, and more efficiently than ever before.




